
Sahara Hot Start PCR Master Mix
Highlights
- Extreme thermal stability: withstands three months at 25 ºC with no impact on efficiency
- Aptamer-based hot start system reduces non-specific amplification and primer dimers
- Overcomes inhibition in crude extracts and environmental samples
- Amplifies up to 4 kb from genomic DNA, and GC-rich targets with up to 70% GC-content
- License-free for commercial or diagnostic use
Description
Sahara Hot Start PCR Master Mix is a high-efficiency 2X Taq mix ideal for endpoint PCR, genotyping, and cloning applications, as well as the quantitative amplification of singleplex qPCR targets using probes.
The Most Stable Master Mix on the Planet
Stable at 50 ºC for 8 days, or 25 ºC for 3 months, Sahara mixes ship economically at ambient temperatures, prevent Cq drift over time due to thermal degradation, and are ideal for high throughput or field applications where temperature control is not always ideal.
High Efficiency and Clear Gel Bands
Set up reactions with ease at room temperature: an aptamer-based hotstart system prevents amplification from occurring below 50 ºC, reducing the non-specific amplification and primer-dimers which harm efficiency and produce blurry gels.
Amplifies Everything Without Inhibition
Sahara Hot Start has been validated against genomic, plasmid, and cDNA targets from a wide range of organisms. The mix supports both standard and fast protocols, and amplifies GC-rich targets up to 70%.
Numerous enhancers bind & inactivate PCR inhibitors, allowing amplification from crude extracts of:
- Blood
- Food & beverages
- Plant & animal tissue
- Environmental air & water samples
Performance
High Efficiency & Linearity
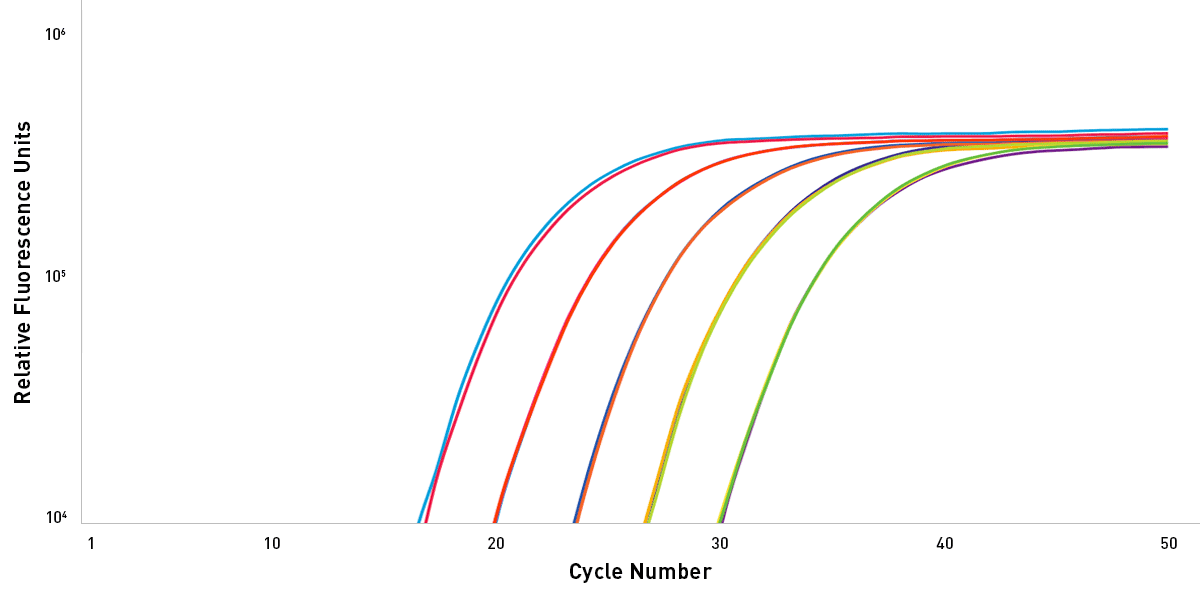
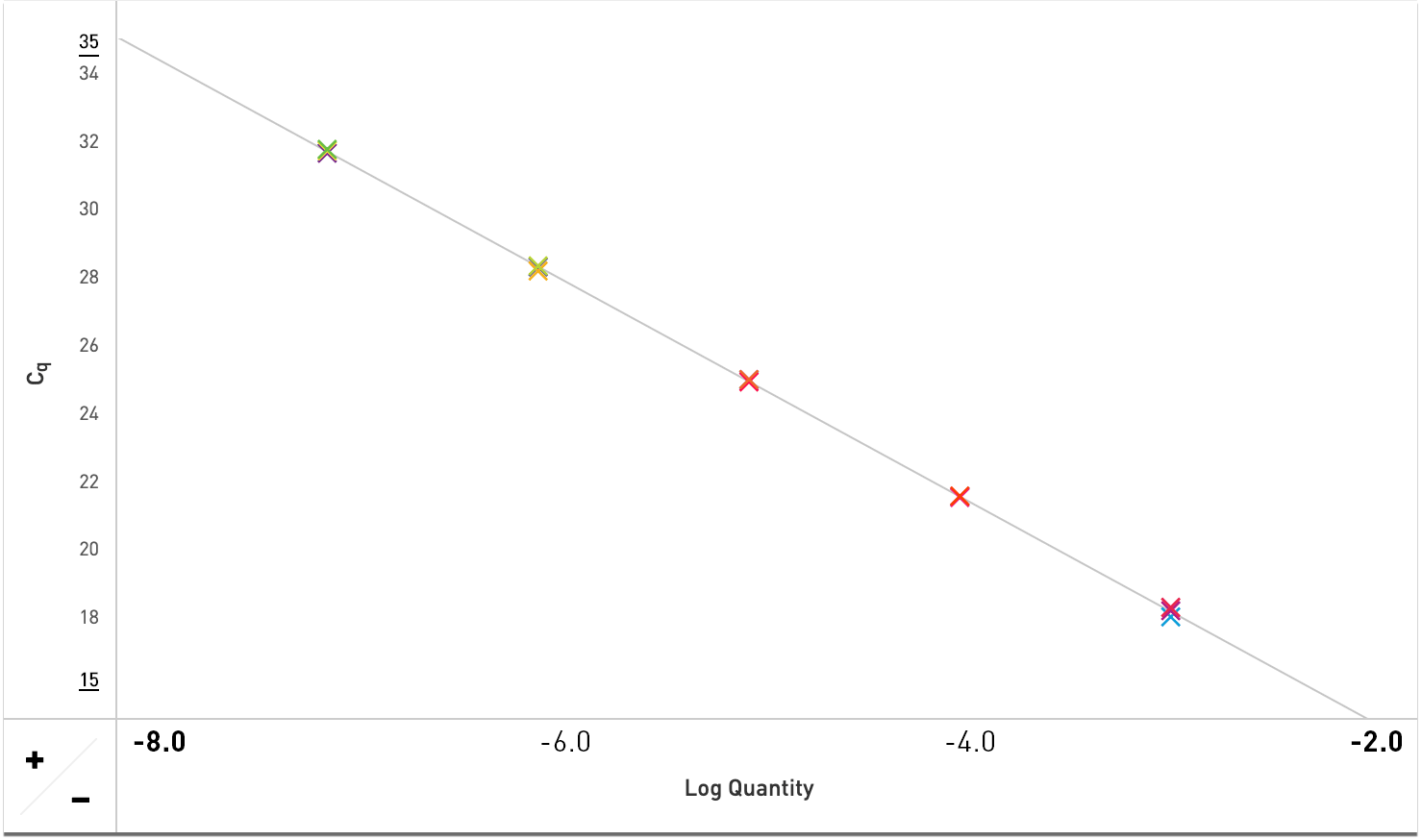
Exceptional efficiency & linearity is demonstrated using Sahara Hot Start PCR Master Mix to amplify a 157 bp gene fragment on an Open qPCR instrument in this 5-point standard curve.
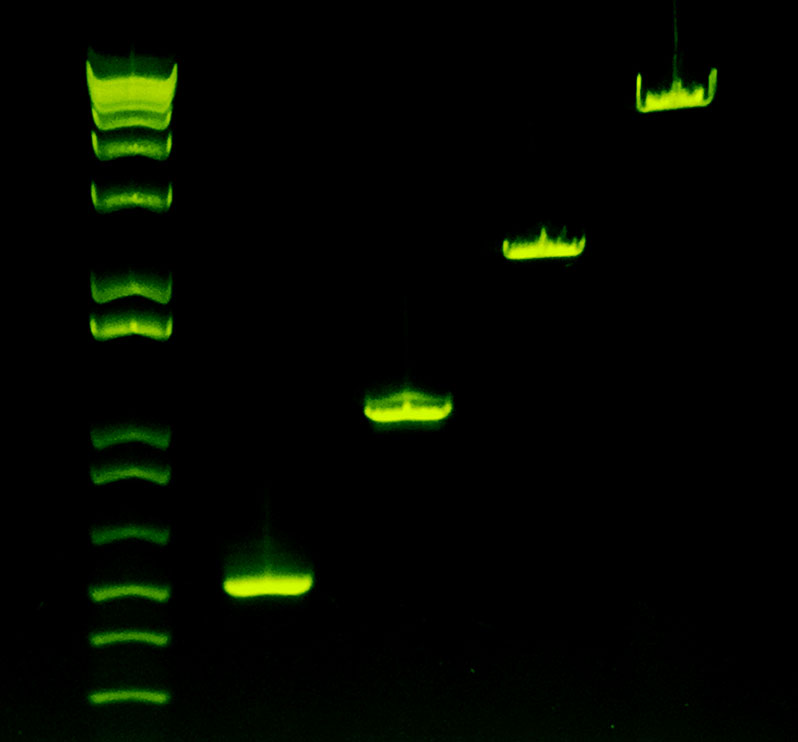
Amplifies Everything
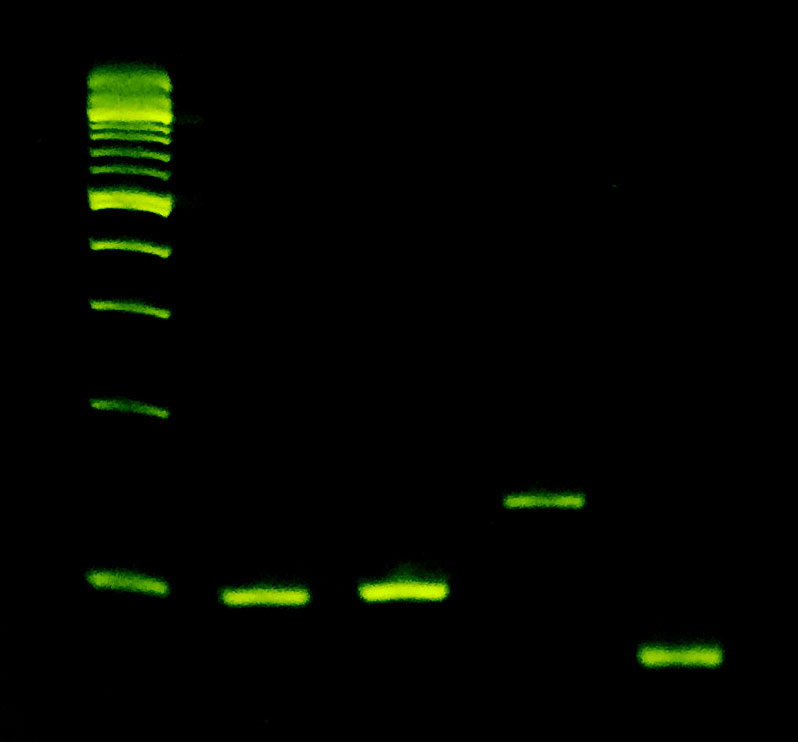
Left: HPRT (42% GC), Ara h2 (51% GC), hActin3 (62% GC), and Salmon Sperm (67% GC) targets, ranging from 77 – 172 bp, were amplified on an Open qPCR instrument with Sahara Hot Start. All targets resolved well.
Right: Lambda phage targets of 500 bp, 1000 bp, 1978 bp, and 4003 bp show consistent amplification using Sahara Hot Start.
Incredibly Stable
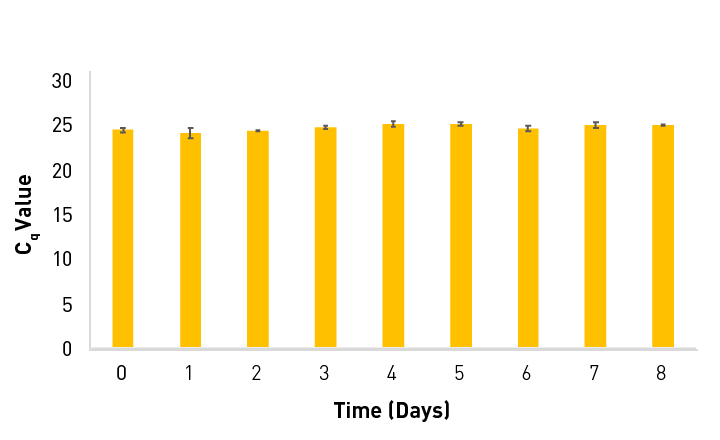
Sahara Hot Start PCR Master Mix was stored at 50 ºC, and a singleplex qPCR reaction was run daily on the Open qPCR instrument. No difference in Cq is observed throughout the trial.
Fast & Effective Hot Start
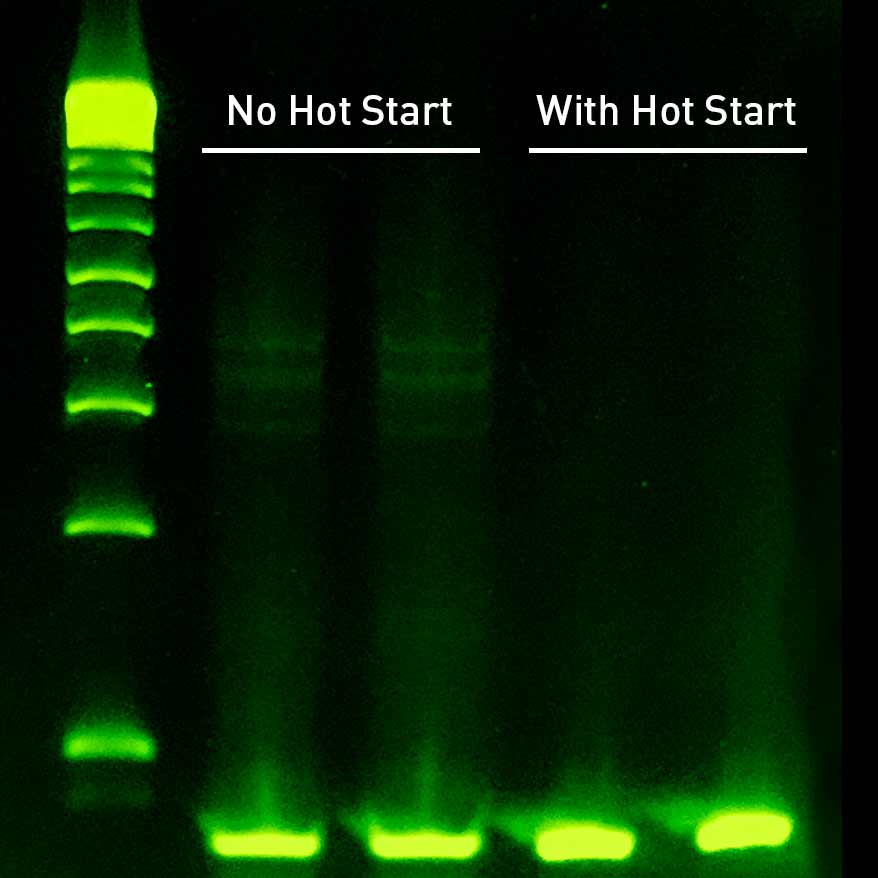
A challenging salmon sperm assay with 67% GC content was run with and without the Sahara Hot Start aptamer. The aptamer hot start successfully prevents the non-specific products seen in the non-hot start assay.
Specifications
PCR
| Concentration | 2X |
| Hot Start Mechanism | Aptamer |
| GC Content | 40 – 70% |
| Reaction Speed | Fast |
| Max Amplicon Length | 4 kb |
| Exonuclease Activity | 5' → 3' |
| Supported Probes | TaqMan/Hydrolysis, Molecular Beacon, Scorpions |
| Supported Templates | Genomic DNA, cDNA, Plasmid DNA |
| Recommended Reaction Volume | 10 – 50 µL |
Shipping, Storage, and Stability
| High Temperature Stability | 8 days @ 50 ºC, 3 months @ 25 ºC |
| Freeze/Thaw Cycles | Up to 20 |
| Shipping Conditions | Ambient |
| Storage Conditions | -20 ºC long-term, or 4 ºC for 6 months |
Compatible Instruments
The mix may be used with all endpoint as well as no-ROX qPCR instruments. It may be used with high and low-ROX qPCR instruments if a ROX reference dye is added by the user.
FAQs
What is a PCR Master Mix?
A PCR master mix is a premixed solution that contains most of the components necessary to run a PCR assay. The mix contains Taq DNA polymerase, dNTPs, MgCl2, as well as enhancers and stabilizers in a buffer that is optimized for DNA amplification by PCR. Read more...
License
To promote collaboration in science, Sahara Hot Start PCR Master Mix is provided under an open license:
- It may be used freely for any application, including commercial and diagnostic use
- No license agreements or royalty payments required for OEM use
- May be incorporated into your own test kits without restriction
Resources
Certificates of Analysis
| View Sample Certificate |
